This
article talks about marine corrosion, mainly in regard to small
commercial and recreational craft; what causes this corrosion to
take place and what reasonable steps can be instigated to prevent
or minimize this corrosion. Much of what is discussed here
can be applied to other structures operating in sea water.
As
the title implies, this is only a quite basic introduction to what
is a complex and varied subject. This article is intended only for
those people who do not have a technical background in corrosion
but may have to deal with its consequences. We have
provided a set of references at the end of this article, which provide
more in depth material.
So…what
is corrosion?
Corrosion
is an electrochemical process
involving metals. That
is… electricity and chemical reactions are involved and metal
material is lost. A common experience for all of us are steel items
rusting. For the "iron post in the ground" diagram below,
area A and C are wet (it has just rained) and electrons are traveling
through the metal from A to C. Area A is losing iron ions
into the water. At area C, due to the presence of oxygen in the
water, and a surplus of electrons
in the metal, water molecules are combining with oxygen atoms and
electrons to produce hydroxyl ions. Ions are positively or
negatively charged atoms or molecules. Positively charged ions are
attracted to negatively charged ions.
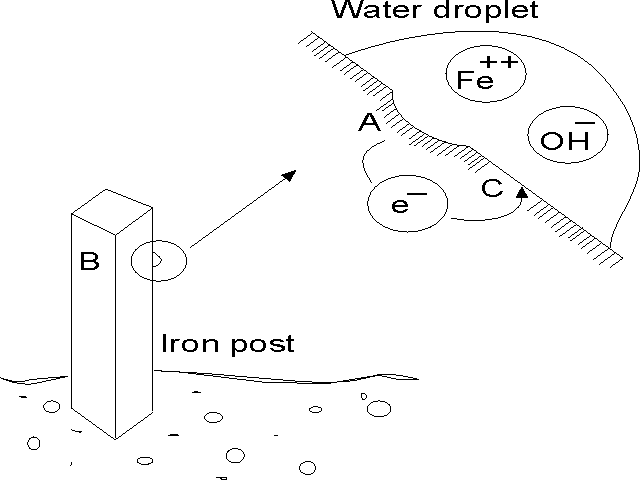
If
they can travel towards one another, such as through the water (in
this case termed the electrolyte),
they will. In the case of the steel and hydroxyl ions, they combine
to form rust. The "iron post in the ground" has corrosion
occurring even though it is one metal.
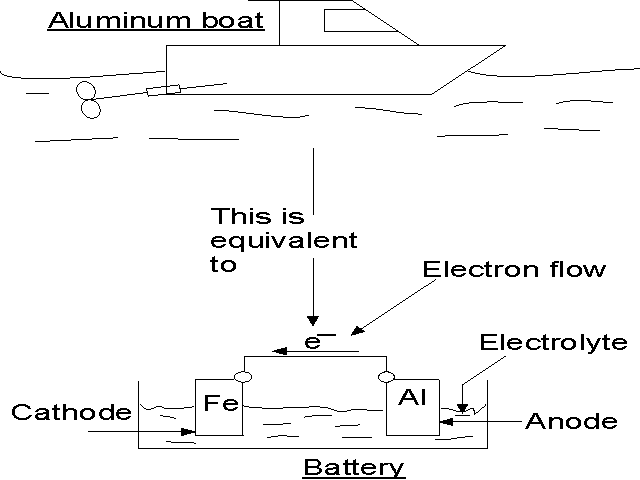
Corrosion
can occur quite rapidly when dissimilar metals are electrically
connected together. The next picture shows an aluminum hull and
stainless steel propeller immersed in an electrolyte (seawater).
If the propeller and hull are electrically connected, you have a
battery with electrical current flowing. The corrosion associated
with the connection of two dissimilar metals is called galvanic
corrosion. The aluminum is losing material in the
form of aluminum ions. Electrons are flowing from the aluminum through
the electrical connection to the stainless steel. The metal supplying
the electrons is called the anode
and the metal receiving them is called the cathode.
For
galvanic corrosion to occur, there must be an electrical circuit.
This means there must be a path for the electrons to travel between
the two dissimilar metals and there must be a path through the electrolyte
for the ions to travel. Additionally, the chemical reaction at the
cathode usually requires the presence of oxygen in the electrolyte.
The
photo below shows a stainless steel bolt passing through an aluminum
casting (part of an out drive). Pitting corrosion is evident on
the aluminum with the stainless steel bolt in the virtually 'as
new' condition.

Galvanic
series
Graphite
----------------------------- +200 to +300 mV
Stainless
steel (passive) ---------------
0 to –100 mV
Bronze
---------------------------------------- –240 to –310
mV
Copper
---------------------------------------- –300 to –570
mV
Brass
------------------------------------------ –300 to –400
mV
Mild
steel ------------------------------------- –600 to –710
mV
Aluminum
------------------------------------- –740 to –980 mV
Zinc
---------------------------------------------- -980 to –1030
mV
Magnesium
----------------------------------- –1500 to –1700 mV
Your
boat will almost certainly have a mix of metals such as steel, aluminum,
copper alloys and so on. Typical metals are listed in order of their
activity in the Table above. For example, electrically connecting
bronze to aluminum in a common electrolyte such as seawater will
result in the aluminum being the anode and thus corroding. How do
you protect an aluminum hull? Looking at the Table: zinc is more
active than aluminum. Hence it is usual to fit zinc anodes on a
hull to protect the aluminum. The zinc corrodes in preference to
aluminum.
An
example of a good zinc anode and a 'bad' zinc anode is shown adjacent
in the photo. Both zincs are trying to protect the aluminum pump
housing. Which zinc is working correctly? Answer: the one on the
left which is corroding. It is a common fallacy that if one zinc
lasts longer than another (given the same component to protect and
the same size anodes) then it is a better zinc. The reverse is true.
The zinc on the right in the photo either has a poor electrical
contact between the zinc and the aluminum, or it has been contaminated
with iron (50 parts per million is enough) during the manufacturing
process.

How
do you check if an anode is working? If the boat is in the water,
then looking to see if the anode is corroding is one way. This is
not completely fool proof as the anode may still be functioning
correctly but the protection level may be insufficient to prevent
corrosion.
A good
way to check if the protection level adequate is to use a reference
electrode and a good quality multimeter set to DC volts. The reference
electrode terminal is plugged into the COM plug on the multimeter.
See the diagram below where an aluminum hull is fitted with zinc
anodes. The out drive is also electrically connected to the hull.
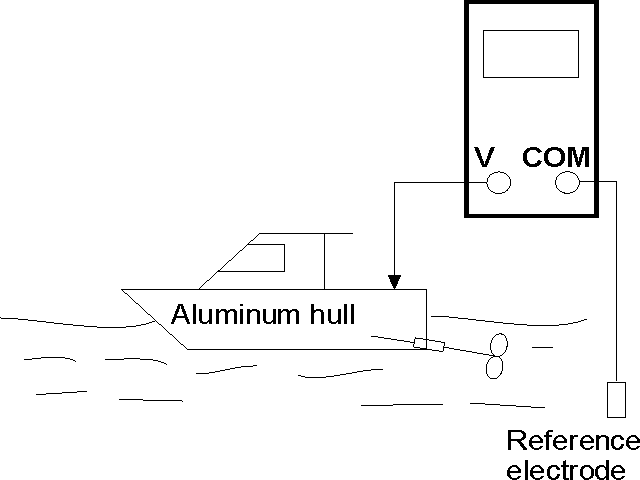
Place
the reference electrode (silver silver chloride) in the sea water,
and connect the multimeter red lead to the hull. If the reading
on the multimeter is between -850 mV and -1100 mV, then the zincs
are working and your boat hull is protected. You should then repeat
the test with the out drive.
Hot
docks
Some
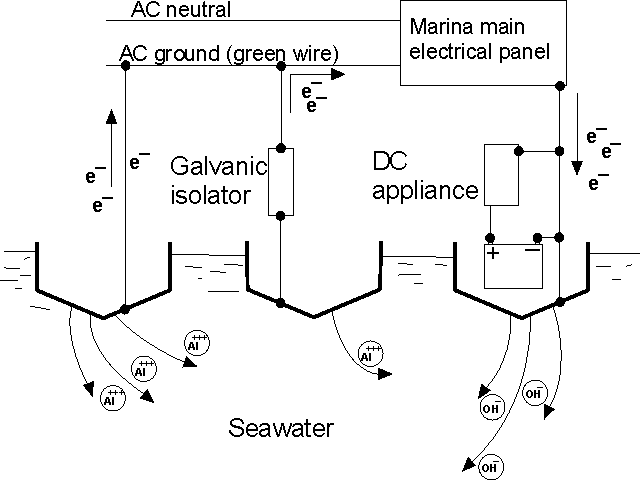
marinas have a reputation as a 'hot dock'. The term arises from
boats seemingly experiencing more severe corrosion when visiting
a particular dock. Often the cause is small DC voltages present
in the green neutral wire on the shore power point. The diagram
below illustrates the problem. Three boats are connected to the
shore power. One boat has a DC electrical appliance which is faulting
to battery positive. This results in a positive bias on the boat
neutral which attracts electrons from the other boats. Boat A experiences
severe corrosion. Boat B has a galvanic isolator fitted. If the
DC voltages are low (say under 1 volt) the galvanic isolator will
prevent DC current from flowing and will protect Boat B.
Galvanic
isolator
The
galvanic isolator is a relatively simple device which generally
contains a pair of diodes and a capacitor. A galvanic isolator
should prevent small DC currents that may be present on the shore
power neutral wire from causing corrosion on your boat. An alternative
to a galvanic isolator is an isolation transformer. ABYC standards
(USA) set guidelines. Not using a galvanic isolator (or isolation
transformer) and connecting to shore power is a bit like playing
Russian roulette. You are taking a chance and you may get away with
it for now...........
Corrosion
Sentry
For
peace of mind and protection of your investment, we have developed
an intelligent corrosion monitor. This monitor comes in three variants:
Sentry
Further
reading
Everything
you need to know about corrosion, Quicksilver Marine
Parts and Accessories
Corrosion,
Vol 1 Metal Environment reactions,
edited by Sheir L L, Jarman R A, Burstein G T, Butterworth Heinmann,
1998
Corrosion,
Vol 2, Corrosion control, edited
by Sheir L L, Jarman R A, Burstein G T, Butterworth Heinmann, 1998
